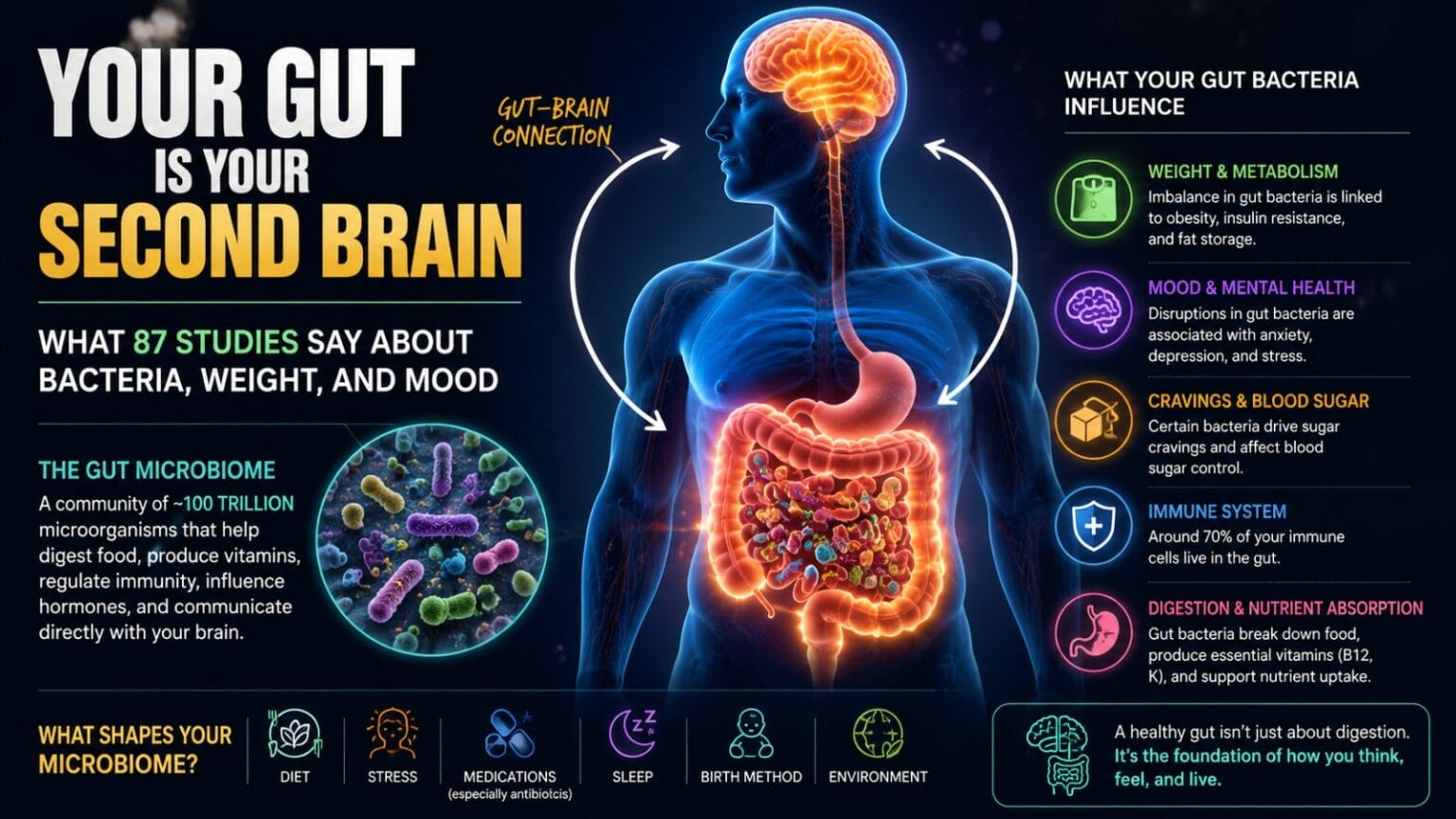
What if the root cause of your weight gain, your low energy, your anxiety, and your sugar cravings wasn’t willpower — but bacteria? That’s not a wellness influencer’s hot take. It’s the emerging consensus of thousands of peer-reviewed studies, including a landmark review of 87 studies that directly links gut bacteria to obesity, insulin resistance, and mood disorders.
In 2026, gut health has moved from the fringes of wellness culture into the centre of mainstream medicine. Gastroenterologists, psychiatrists, endocrinologists, and nutritionists are all now looking at the same thing: the 100 trillion microorganisms living in your digestive tract — and what happens when they go wrong.
This is everything you need to know.
What Is the Gut Microbiome?
Your gut microbiome is the vast ecosystem of bacteria, fungi, viruses, and other microorganisms that live in your digestive system — primarily in your large intestine. The human gut contains approximately 100 trillion microbial cells, outnumbering human cells in your body by a ratio of roughly 1.3 to 1.
These microorganisms are not passengers. They are active participants in almost every system in your body. They help digest food, produce vitamins, regulate your immune system, influence your hormones, and — as research is now confirming — directly communicate with your brain.
The composition of your microbiome is as unique as your fingerprint, shaped by your diet, environment, stress levels, medications, sleep patterns, and even the manner of your birth. And when that composition falls out of balance — a state scientists call dysbiosis — the downstream effects are profound.
Also read – Digital Health is a Gold rush
The Gut–Obesity Connection: What the Research Shows
The link between gut bacteria and body weight is one of the most actively researched areas in modern medicine — and the evidence is compelling.
A comprehensive review published in 2025 in a leading peer-reviewed journal examined the relationship between the gut microbiome and human obesity across thousands of patients. The findings confirm that people with obesity consistently show reduced microbial diversity in their gut compared to lean individuals — fewer species, less variety, and a higher ratio of certain bacterial families associated with fat storage and inflammation.
Here is the mechanism: gut bacteria regulate energy balance and insulin sensitivity via short-chain fatty acids, induce chronic inflammation, modulate metabolic and appetite genes, alter bile acid signalling, and promote fat storage by inhibiting a compound called fasting-induced adipose factor. In other words, your gut bacteria don’t just respond to what you eat — they actively influence how your body processes and stores it.
Researchers from the METACARDIS project gathered data from the gut microbiomes of 2,000 people over six years, examining over 1,400 clinical, biological, and lifestyle variables in an attempt to find biomarkers and risk factors for cardiometabolic diseases including obesity. Their findings suggest that a poor nutritional environment and poor lifestyle choices promote the progression of these diseases through changes in gut microbiota — and that healthier diets can lead to improved cholesterol, inflammatory markers, and weight loss through increased gut microbial diversity.
The implication is significant: if you have been struggling with your weight despite eating reasonably and exercising, your microbiome may be working against you — and targeting it directly could be one of the most powerful interventions available.
Gut Bacteria and Insulin Resistance: The Hidden Connection
Insulin resistance — the condition in which your cells stop responding effectively to insulin, causing blood sugar to remain elevated — is the driving force behind Type 2 diabetes, metabolic syndrome, and a cascade of related health conditions. It affects hundreds of millions of people worldwide, many of whom don’t know they have it.
What most people don’t know is that gut bacteria play a direct role in whether or not insulin resistance develops.
Whole bacteria, their products, and metabolites undergo increased translocation through the gut epithelium into circulation due to degraded tight junctions and increased intestinal permeability — a phenomenon sometimes called “leaky gut” — which culminates in inflammation and insulin resistance. When the gut barrier is compromised, bacterial toxins enter the bloodstream, triggering a chronic low-grade inflammatory response that progressively impairs insulin signalling throughout the body.
Microbiome-based therapeutics, including probiotics, synbiotics, and fecal microbiota transplantation, have demonstrated potential in modulating key metabolic and inflammatory pathways associated with obesity and insulin resistance. Early clinical trials are producing promising results, and the integration of microbiome therapies into standard metabolic care is being described by researchers as “increasingly feasible and therapeutically transformative.”
This is not future medicine. Trials are underway right now exploring whether transplanting gut bacteria from metabolically healthy donors into people with insulin resistance and Type 2 diabetes can reverse the condition.
The Gut–Brain Axis: How Your Stomach Controls Your Mood
Here is where the science becomes genuinely extraordinary.
Your gut and your brain are in constant, direct communication through a network of nerves, hormones, and immune signals collectively called the gut–brain axis. This bidirectional communication system connects your gastrointestinal tract to your central nervous system through the vagus nerve — the longest nerve in the body — as well as through hormonal and immune pathways.
The gut microbiome is increasingly recognised as a key modulator of central nervous system function through the gut–brain axis. Evidence indicates that gut microbiota modulate neurochemical pathways involving serotonin, dopamine, GABA, and glutamate, as well as immune and endocrine axes.
Let that land for a moment. The bacteria in your gut are producing and regulating the very neurotransmitters that determine whether you feel calm or anxious, motivated or depressed, focused or foggy.
Approximately 90% of the body’s serotonin — the neurotransmitter most associated with mood, wellbeing, and emotional stability — is produced in the gut, not the brain. When gut bacteria are healthy and diverse, serotonin production is stable. When dysbiosis occurs, serotonin metabolism is disrupted, and mood disorders often follow.
Dysbiosis has been associated with neuropsychiatric disorders including depression, anxiety, impulsivity, cognitive decline, and addiction.
Recent research highlights the association between gut dysbiosis and psychiatric disorders including anxiety, depression, and stress-related conditions. Key findings indicate that altered microbial diversity, decreased short-chain fatty acid production, and increased neuroinflammation contribute to mental health disturbances.
Obesity, Mood Disorders, and the Gut: A Three-Way Relationship
One of the most significant recent findings in this field is the discovery that obesity, mood disorders, and gut dysbiosis form a three-way feedback loop — each condition worsening the others.
Obesity is a complex health condition that not only increases susceptibility to cardiovascular diseases and diabetes, but also has a profound impact on the patient’s mental state, leading to the onset of various mental disorders, particularly mood disorders. The gut microbiota is vital in regulating and maintaining host physiology, including metabolism and neuronal circuits.
In other words, obesity disrupts the gut microbiome, which then disrupts neurotransmitter production, which then worsens mood — which in turn drives emotional eating, poor sleep, and reduced physical activity, which further disrupts the microbiome. Breaking this cycle requires addressing all three components together.
Dietary interventions, prebiotics, probiotics, and fecal microbiota transplantation have yielded encouraging results in addressing the gut-brain axis in mood disorders, though significant advances are still needed to establish next-generation approaches that precisely target the neurobiological mechanisms involved.
Researchers are now calling for the integration of microbiota research directly into psychiatric practice — recognising that treating depression and anxiety without addressing gut health may be leaving the most important lever untouched.
What Disrupts Your Gut Microbiome?
Understanding what harms the microbiome is the first step toward protecting it. The most common disruptors include:
Ultra-processed food. A diet high in refined sugar, artificial sweeteners, and ultra-processed ingredients rapidly reduces microbial diversity and feeds harmful bacterial species over beneficial ones.
Antibiotics. While lifesaving in acute situations, antibiotics are indiscriminate — they kill beneficial bacteria alongside harmful ones. A single course of antibiotics can alter the microbiome for months. Repeated courses may cause permanent changes.
Chronic stress. The gut–brain axis works in both directions. Chronic psychological stress alters gut motility, increases intestinal permeability, and shifts microbial composition toward inflammatory species. Stress and gut health are inseparable.
Poor sleep. Circadian rhythm disruption — irregular sleep patterns, shift work, late-night eating — is now known to negatively alter gut microbiome composition. Sleep and the microbiome are in a bidirectional relationship: each depends on the other.
Sedentary lifestyle. Physical activity promotes microbial diversity. People who exercise regularly have more varied and resilient gut microbiomes than those who are sedentary — independent of diet.
Alcohol. Excessive alcohol consumption damages the gut lining, increases intestinal permeability, and reduces populations of beneficial bacteria.
How to Improve Your Gut Health: Evidence-Based Strategies
The good news is that the microbiome is remarkably responsive to change. Here is what the evidence supports:
Eat more fermented foods. Yoghurt, kefir, kimchi, sauerkraut, miso, and kombucha are rich in live bacterial cultures that directly replenish beneficial gut populations. A landmark Stanford study found that a diet high in fermented foods increased microbial diversity and reduced inflammatory markers in just ten weeks.
Increase dietary fibre. Fibre is the primary food source for beneficial gut bacteria. Whole grains, legumes, vegetables, fruits, nuts, and seeds all feed the bacteria that produce short-chain fatty acids — the compounds responsible for gut lining integrity, insulin sensitivity, and anti-inflammatory signalling.
Eat a diverse plant-based diet. Research suggests that eating 30 or more different plant foods per week is associated with significantly higher gut microbial diversity. Variety, not just quantity, is the key principle.
Take a quality probiotic. Modulating gut microbiota through probiotics, prebiotics, diet, and fecal microbiota transplantation is emerging as a potential therapeutic strategy for both metabolic and mental health conditions. Several clinical studies suggest that interventions targeting gut microbiota may alleviate symptoms of depression and anxiety.
Manage stress actively. Mindfulness, breathwork, regular exercise, and adequate sleep all have demonstrated positive effects on the gut microbiome — through the same gut–brain axis that connects them.
Limit ultra-processed food. This is perhaps the single most impactful dietary change most people can make. Removing highly processed foods from your diet creates the conditions for beneficial bacteria to flourish within weeks.
The Future of Gut Health: Personalised Microbiome Medicine
The frontier of gut health research is moving rapidly toward personalisation. Mechanisms like tryptophan metabolism, the HPA axis, and microbial metabolites such as short-chain fatty acids are being studied as links between diet, gut microbiota, and depression — with clinical trials showing promising effects of probiotics in alleviating depressive symptoms.
Within the next decade, personalised microbiome analysis — a stool test that maps your unique microbial ecosystem and recommends targeted dietary and probiotic interventions — is expected to become a standard part of preventive healthcare. Companies are already offering consumer versions of this technology today, with clinical-grade versions not far behind.
The gut microbiome may prove to be the most important biomarker of overall health that medicine has yet to fully integrate into routine care.
The Bottom Line
Your gut is not just a digestive organ. It is a second brain, an immune headquarters, a metabolic regulator, and a mood stabiliser — all in one. The 100 trillion organisms living inside it are not incidental to your health. They are central to it.
The research is now unambiguous: the state of your gut microbiome directly influences your weight, your metabolic health, your risk of insulin resistance, and your mental wellbeing. Eighty-seven studies — and counting — confirm the connection.
You cannot out-supplement a bad diet, out-exercise chronic stress, or out-medicate a disrupted microbiome indefinitely. But you can feed your gut well, diversify your plant intake, manage your stress, sleep properly, and move your body — and watch what happens.
Start with your gut. Everything else follows.
Subscribe to our YouTube Channel The Logic Stick for more video insights
Sources: NCBI / PubMed | METACARDIS Research Project | University of Cassino — Nutrients Journal (2025) | Cureus Medical Journal (2025) | MDPI Life Journal (2025) | University of California San Francisco | University of Parma (2025) | Rock Health | Prenuvo Health Blog | DrAxe.com